803 Restriction — When Proper [R-07.2022]
Under the statute, the claims of an application may properly be required to be restricted to one of two or more claimed inventions only if they are able to support separate patents and they are either independent (MPEP § 802.01, § 806.06, and § 808.01) or distinct (MPEP § 806.05 - § 806.05(j)).
If the search and examination of all the claims in an application can be made without serious burden, the examiner must examine them on the merits, even though they include claims to independent or distinct inventions.
I. CRITERIA FOR RESTRICTION BETWEEN PATENTABLY DISTINCT INVENTIONSThere are two criteria for a proper requirement for restriction between patentably distinct inventions:
- (A) The inventions must be independent (see MPEP § 802.01, § 806.06, § 808.01) or distinct as claimed (see MPEP § 806.05 - § 806.05(j)); and
- (B) There would be a serious search and/or examination burden on the examiner if restriction is not required (see MPEP § 803.02, § 808, and § 808.02).
Examiners must provide reasons and/or examples to support conclusions, but need not cite documents to support the restriction requirement in most cases.
Where plural inventions are capable of being viewed as related in two ways, both applicable criteria for distinctness must be demonstrated to support a restriction requirement.
If there is an express admission that the claimed inventions would have been obvious over each other within the meaning of 35 U.S.C. 103, restriction should not be required. In re Lee, 199 USPQ 108 (Comm’r Pat. 1978).
For purposes of the initial requirement, a serious search burden on the examiner may be prima facie shown by appropriate explanation of separate classification, or separate status in the art, or a different field of search as defined in MPEP § 808.02. Similarly, a serious examination burden, for example, may be prima facie shown by appropriate explanation of non-prior art issues under 35 U.S.C. 101, pre-AIA 35 U.S.C. 112, first paragraph, and/or 35 U.S.C. 112(a) relevant to one invention that are not relevant to the other invention. A prima facie showing of serious search and/or examination burden may be rebutted by appropriate showings or evidence by the applicant. Insofar as the criteria for restriction practice relating to Markush claims is concerned, the criteria is set forth in MPEP § 803.02. Insofar as the criteria for restriction or election practice relating to claims to genus-species, see MPEP § 806.04 - § 806.04(i) and § 808.01(a).
803.01 Review by Examiner with at Least Partial Signatory Authority [R-07.2015]
Since requirements for restriction under 35 U.S.C. 121 are discretionary with the Director, it becomes very important that the practice under this section be carefully administered. Notwithstanding the fact that this section of the statute apparently protects the applicant against the dangers that previously might have resulted from compliance with an improper requirement for restriction, IT STILL REMAINS IMPORTANT FROM THE STANDPOINT OF THE PUBLIC INTEREST THAT NO REQUIREMENT BE MADE WHICH MIGHT RESULT IN THE ISSUANCE OF TWO PATENTS FOR THE SAME INVENTION. See MPEP § 804.01. Therefore, to guard against this possibility, only an examiner with permanent full signatory authority or temporary full signatory authority may sign final Office actions containing a final requirement for restriction. An examiner with permanent partial signatory authority or temporary partial signatory authority may sign non-final Office actions containing a final requirement for restriction.
803.02 Election of Species Requirements – Markush Claims [R-07.2022]
I. MARKUSH CLAIMSA “Markush” claim recites a list of alternatively useable members. In re Harnisch, 631 F.2d 716, 719-20, 206 USPQ 300, 303 (CCPA 1980); Ex parte Markush, 1925 Dec. Comm'r Pat. 126, 127 (1924). The listing of specified alternatives within a Markush claim is referred to as a Markush group or a Markush grouping. Abbott Labs v. Baxter Pharmaceutical Products, Inc., 334 F.3d 1274, 1280-81, 67 USPQ2d 1191, 1196 (Fed. Cir. 2003)(citing to several sources that describe Markush groups).
When examining a Markush claim, the examiner may generally choose to require a provisional election of species from among patentably indistinct species or patentably indistinct groups of species. See subsection III, below. The applicant’s election serves as a starting point for the search and examination of the claim.
See MPEP § 2117 for a general discussion of Markush claims, guidance and examples regarding the determination of whether a Markush grouping is proper, and rejections on the basis that a claim contains an improper Markush grouping. A rejection based on an improper Markush grouping should be made in an Office action on the merits. In certain circumstances, both a provisional election of species requirement and an improper Markush grouping rejection may apply to the same claim.
See MPEP § 2111.03, subsection II, and MPEP § 2173.05(h) for a discussion of Markush claims and compliance with the definiteness requirement of 35 U.S.C. 112(b).
II. PROPER MARKUSH GROUPINGPursuant to the Supplementary Examination Guidelines for Determining Compliance with 35 U.S.C. 112 and for Treatment of Related Issues in Patent Applications (“Supplementary Guidelines”), 76 Fed. Reg. 7162 (February 9, 2011), a Markush grouping is proper if: (1) the members of the Markush group share a “single structural similarity,” and (2) the members share a common use. Id. (citing In re Harnisch, 631 F.2d 716, 721-22, 206 USPQ 300, 305 (CCPA 1980)).
Where a Markush grouping describes part of a combination or process, the members following “selected from the group consisting of” (or a similar introductory phrase) must be substitutable, one for the other, with the expectation that the same intended result would be achieved. Multilayer Stretch Cling Film Holdings, Inc. v. Berry Plastics Corp., 831 F.3d 1350, 1357, 119 USPQ2d 1773, 1779 (Fed. Cir. 2016)(“It is generally understood that … the members of the Markush group … are alternatively usable for the purposes of the invention … .”)(citations omitted). Where a Markush grouping describes part of a chemical compound, regardless of whether the claim is limited to a compound per se or the compound is recited as part of a combination or process, the members following “selected from the group consisting of” (or similar introductory phrase) need not share a community of properties themselves; the propriety of the grouping is determined by a consideration of the compound as a whole. See Harnisch, 631 F.2d at 722, 206 USPQ at 305 (“in determining the propriety of a Markush grouping the compounds must be considered as wholes and not broken down into elements or other components”).
See MPEP § 2117 for guidance and examples regarding the determination of whether a Markush grouping is proper.
In accordance with the principles of compact prosecution, if the examiner determines that one or more claims appear to include an improper Markush grouping (see MPEP § 2117), the examiner should require the applicant to elect a species. Note that if a written provisional election of species requirement must be made separate from the first Office action on the merits, it should not include a rejection on the basis of an improper Markush grouping. Any appropriate improper Markush grouping rejection should be made in an Office action on the merits.
III. ELECTION OF SPECIES PRACTICE FOR MARKUSH CLAIMSA. OverviewMarkush claims recite a plurality of alternatively usable substances or members. In most cases, a recitation by enumeration is used because there is no appropriate or true generic language. A Markush claim may include independent and distinct inventions. This is true where two or more of the members are so unrelated and diverse that a prior art reference anticipating the claim with respect to one of the members would not render the claim obvious under 35 U.S.C. 103 with respect to the other member(s). In applications containing a Markush claim that encompasses at least two independent or distinct inventions, the examiner may require a provisional election of a single species (or grouping of patentably indistinct species) prior to examination on the merits, with one exception. If the members of a proper Markush group are sufficiently few in number or so closely related that a search and examination of the entire claim can be made without serious burden, the examiner must examine all the members of the Markush group in the claim on the merits, even though they may be directed to independent and distinct inventions. In such a case, the examiner will not require provisional election of a single species. See MPEP § 808.02
An election of species requirement is a type of restriction requirement. An examiner should set forth a requirement for election of a single disclosed species (or a grouping of patentably indistinct species) in a Markush claim using form paragraph 8.01 when claims limited to species are present or using form paragraph 8.02 when no species claims are present. See MPEP § 808.01(a) and § 809.02(a). If a Markush claim depends from or otherwise requires all the limitations of another generic or linking claim, see MPEP § 809.
Following election, the Markush claim will be examined fully with respect to the elected species and further to the extent necessary to determine patentability. Note that where a claim reads on multiple species, only one species needs to be taught or suggested by the prior art in order for the claim to be anticipated or rendered obvious. See, e.g., Fresenius USA, Inc. v. Baxter Int’l, Inc., 582 F.3d 1288, 1298, 92 USPQ2d 1163, 1171 (Fed. Cir. 2009)(the entire element is disclosed by the prior art if one alternative in the Markush group is in the prior art).
If the Markush claim is not allowable, the provisional election will be given effect and examination will be limited to the Markush claim and claims to the elected species, with claims drawn to species patentably distinct from the elected species held withdrawn from further consideration. As an example, in the case of an application with a Markush claim drawn to the compound X-R, wherein R is a radical selected from the group consisting of A, B, C, D, and E, the examiner may require a provisional election of a single species, XA, XB, XC, XD, or XE. The Markush claim would then be examined fully with respect to the elected species and any species considered to be clearly unpatentable over the elected species.
If on examination the elected species is found to be anticipated or rendered obvious by prior art, the Markush claim and claims to the elected species will be rejected, and claims to the nonelected species will be held withdrawn from further consideration.
If the examiner determines that the elected species is allowable over the prior art, the examination of the Markush claim will be extended. If prior art is then found that anticipates or renders obvious the Markush claim with respect to a nonelected species, the Markush claim shall be rejected; claims to the nonelected species would still be held withdrawn from further consideration. The prior art search will not be extended unnecessarily to cover all nonelected species, and need not be extended beyond a proper Markush grouping. See subsection III.C.2, below, for additional guidance.
Should applicant, in response to a rejection of a Markush claim, overcome the rejection by amending the Markush claim to exclude the species anticipated or rendered obvious by the prior art, the amended Markush claim will be examined again. The examination will be extended to the extent necessary to determine patentability of the Markush claim. In the event prior art is found during this examination that anticipates or renders obvious the amended Markush claim, the claim will be rejected and the action can be made final unless the examiner introduces a new ground of rejection that is neither necessitated by applicant’s amendment of the claims nor based on information submitted in an information disclosure statement filed during the period set forth in 37 CFR 1.97(c) with the fee set forth in 37 CFR 1.17(p). See MPEP § 706.07(a). Amendments submitted after the final rejection further restricting the scope of the claim may be denied entry if they do not comply with the requirements of 37 CFR 1.116. See MPEP § 714.13.
B. Provisional Election of SpeciesIf a claim that includes a Markush grouping reads on two or more patentably distinct inventions, a provisional election of species requirement may be made at the examiner’s discretion. When making such a requirement, the examiner will require the applicant to elect a species or group of patentably indistinct species for initial search and examination. The examiner should not require provisional election between species that are not patentably distinct, or when the Markush group is proper and there would be no serious burden if the species were searched and examined together. The examiner should not invite the applicant to elect any group of species that would clearly be rejectable either as an improper Markush grouping or under 35 U.S.C. 112(a) if presented in a separate claim.
In accordance with current practice, when an examiner chooses to require a provisional election of species, in most cases the examiner should call the applicant to request a telephonic election. See MPEP § 812.01. If the applicant elects by telephone, form paragraph 8.23 should be used in the next Office action on the merits. The examiner should note whether the election was made with or without traverse. If a rejection on the basis of an improper Markush grouping is to be made, it should be done in the first Office action on the merits with the written provisional election of species requirement.
If a written provisional election of species requirement is made prior to the first Office action on the merits, it should not include a rejection on the basis of an improper Markush grouping. Any appropriate improper Markush grouping rejection should be made in an Office action on the merits. If during prosecution a new claim is added that includes an improper Markush grouping, or an existing claim is amended to include an improper Markush grouping, the examiner may require provisional election of species at that time, in the same action as any appropriate rejections . Include form paragraph 8.23.01 if the applicant declined to elect by telephone.
C. Initial Examination of Elected Species1. Rejection of Claims to Elected SpeciesExamination on the merits begins after the applicant’s election. If the elected species or group of patentably indistinct species is anticipated by or obvious over the prior art, an appropriate art-based rejection of any claim that reads on the elected species or group of patentably indistinct species should be made. Non-prior art rejections that apply to the elected species or group of patentably indistinct species should also be made. If the election was made with traverse, it should be treated in accordance with MPEP § 821.01.
If the Markush grouping was improper, a rejection on the basis of there being an improper Markush grouping should be made as described in MPEP § 2117. The examiner should use form paragraph 8.40 to make the improper Markush grouping rejection and to advise the applicant of the species that do not belong to a proper Markush grouping that includes the elected species. The form paragraph also serves to advise the applicant that a rejection on the basis of there being an improper Markush grouping is an appealable rather than a petitionable matter.
A claim is drawn to a proper Markush grouping of species A, B, or C. The three species are patentably distinct, and the examiner requires a provisional election. Species A is elected. The examiner rejects species A over prior art, and indicates that species B and C have not been searched and examined. Use form paragraph 8.01 or 8.02 as appropriate to set forth the election requirement.
A claim is drawn to a Markush grouping of species A, B, C, D, or E. The five species are patentably distinct, and the examiner requires a provisional election. The grouping of species A, B, or C is a proper Markush grouping. However, the grouping of species A, B, C, D, or E is not a proper Markush grouping. Species A is elected. The examiner rejects species A over prior art, and indicates that species B, C, D, and E have not been searched and examined. Use form paragraph 8.01 or 8.02 as appropriate to set forth the election requirement. The examiner should also reject the claim on the basis of there being an improper Markush grouping using form paragraph 8.40. The improper Markush grouping rejection should indicate that species D and E do not belong to the proper Markush grouping of species A, B, or C.
If the elected species or group of patentably indistinct species is not anticipated by or obvious over the prior art, the examiner should extend the search and examination to a non-elected species or group of species that falls within the scope of a proper Markush grouping that includes the elected species. The search and examination should be continued until either (1) prior art is found that anticipates or renders obvious a species that falls within the scope of a proper Markush grouping that includes the elected species, or (2) it is determined that no prior art rejection of any species that falls within the scope of a proper Markush grouping that includes the elected species can be made. The examiner need not extend the search beyond a proper Markush grouping. In other words, the examiner need not extend the search to any additional species that do not share a single structural similarity and a common use with the elected species (i.e., do not belong to the same recognized physical or chemical class or to the same art-recognized class and/or do not have a common use and/or do not share a substantial structural feature of a chemical compound and a use that flows from the substantial structural feature). The examiner should continue examination of the Markush claim to determine whether it meets all other requirements of patentability (e.g., 35 U.S.C. 101 and 112, nonstatutory double patenting, and proper Markush grouping).
In the interest of compact prosecution, the examiner should ensure that the record is clear as to which species have been searched and have been found allowable over the prior art. The examiner should indicate that the provisional election of species requirement has been modified if additional species beyond the elected species have been searched and determined to be allowable over the prior art. The examiner should indicate that the provisional election of species requirement has been withdrawn if the full scope of the Markush grouping has been searched and been determined to be allowable over the prior art. Note that the examiner can only make or maintain any restriction requirement if there would be serious search and/or examination burden. Clarity of the record with regard to the provisional election of species requirement is critical to proper application of 35 U.S.C. 121 in later divisional applications.
If a Markush grouping as set forth in a claim is proper and election of species has been required, the examiner must continue to search the species of the claim unless the claim has been found to be unpatentable over prior art. An examiner may not (such as by way of an Ex parte Quayle action or a Notice of Non-Responsive Amendment) seek to require an applicant to limit the scope of a claim that is directed to a proper Markush group to a subset of species that falls within the scope of the claim in the absence of a rejection of the claim for not complying with the requirements for patentability (e.g., 35 U.S.C. 101, 102, 103, and 112, and nonstatutory double patenting).
D. Final Rejection; Finality of Election RequirementAn Office action may be made final if the requirements of MPEP §§ 706.07 - 706.07(b) are met. If a claim in a first application recites a proper Markush grouping that encompasses patentably distinct inventions, an examiner who has required a provisional election of species need not continue to search the claim if the claim is rejected over prior art in a proper final rejection. That is, in this circumstance, the applicant’s election loses its provisional status and is given full effect under 35 U.S.C. 121. Furthermore, if an applicant files a second application that is a divisional application claiming benefit under 35 U.S.C. 120 of the first application, the 35 U.S.C. 121 shield may be applicable. So long as the consonance requirement is met, a claim in the divisional application to a previously non-elected and unexamined embodiment may not be rejected on the ground of non-statutory double patenting over an embodiment examined in the first application. An amendment canceling the rejected species received after final under 37 CFR 1.116 may typically be denied entry on the basis that it would require further consideration and/or search. If the applicant’s provisional election was made with traverse and the requirement has been made final, the applicant may file a petition for review under 37 CFR 1.144. See MPEP §§ 818.01(c) and 818.01(d).
Note that no Markush claim can be allowed until any improper Markush grouping rejection has been overcome or withdrawn (see MPEP § 2117, subsection III), and all other conditions of patentability have been satisfied.
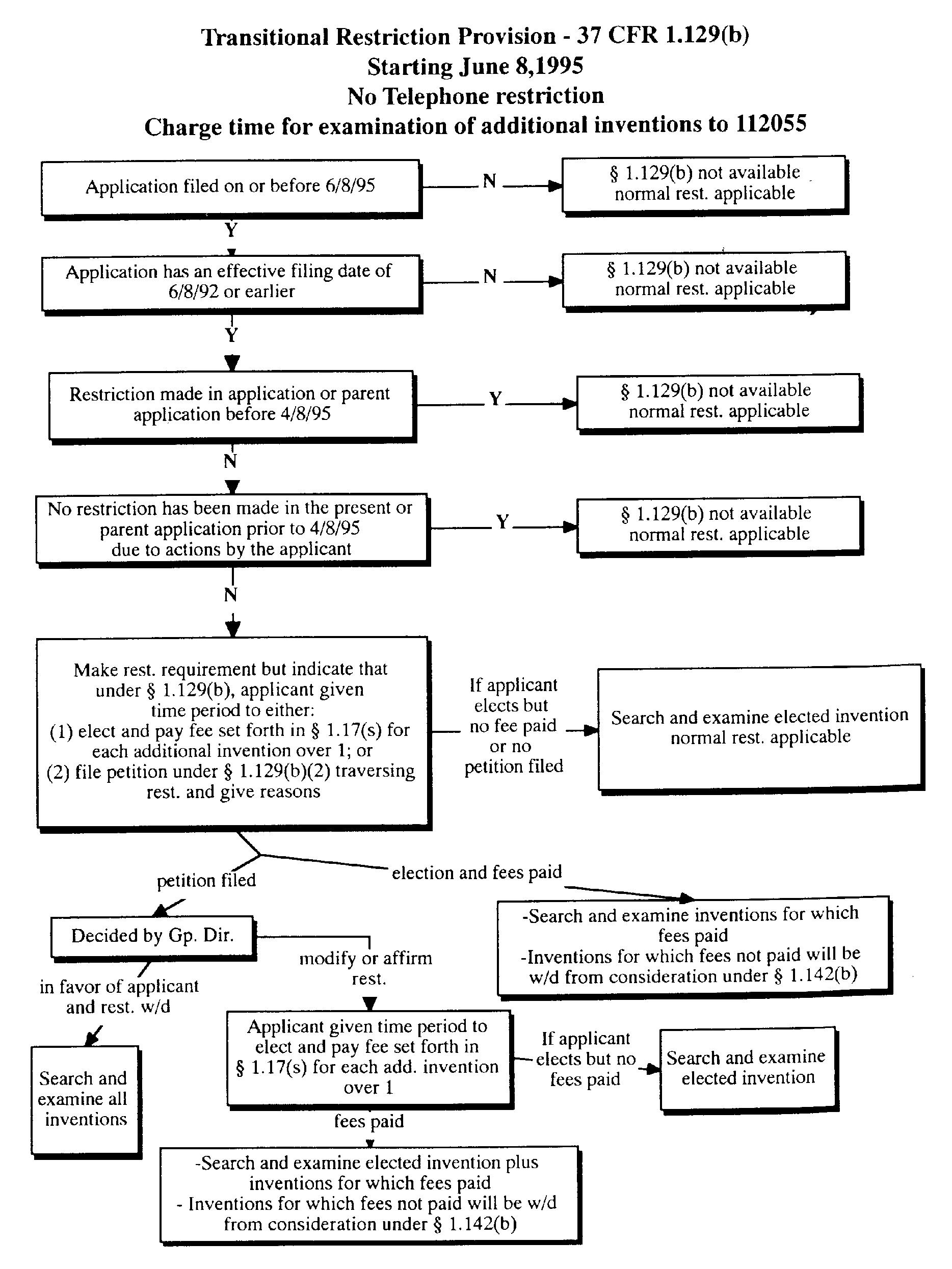
803.03 Transitional Applications [R-08.2012]
PRACTICE RE TRANSITIONAL APPLICATION37 CFR 1.129 Transitional procedures for limited examination after final rejection and restriction practice.
*****
- (b)
- (1) In an application, other than for reissue or a design
patent, that has been pending for at least three years as of June 8,
1995; taking into account any reference made in the application to any
earlier filed application under 35 U.S.C. 120,
121 and 365(c), no
requirement for restriction or for the filing of divisional
applications shall be made or maintained in the application after June
8, 1995, except where:
- (i) The requirement was first made in the application or any earlier filed application under 35 U.S.C. 120, 121, and 365(c) prior to April 8, 1995;
- (ii) The examiner has not made a requirement for restriction in the present or parent application prior to April 8, 1995, due to actions by the applicant; or
- (iii) The required fee for examination of each additional invention was not paid.
- (2) If the application contains more than one independent
and distinct invention and a requirement for restriction or for the
filing of divisional applications cannot be made or maintained
pursuant to this paragraph, applicant will be so notified and given a
time period to:
- (i) Elect the invention or inventions to be searched and examined, if no election has been made prior to the notice, and pay the fee set forth in 1.17(s) for each independent and distinct invention claimed in the application in excess of one which applicant elects;
- (ii) Confirm an election made prior to the notice and pay the fee set forth in § 1.17(s) for each independent and distinct invention claimed in the application in addition to the one invention which applicant previously elected; or
- (iii) File a petition under this section traversing the requirement. If the required petition is filed in a timely manner, the original time period for electing and paying the fee set forth in § 1.17(s) will be deferred and any decision on the petition affirming or modifying the requirement will set a new time period to elect the invention or inventions to be searched and examined and to pay the fee set forth in § 1.17(s) for each independent and distinct invention claimed in the application in excess of one which applicant elects.
- (3) The additional inventions for which the required fee has not been paid will be withdrawn from consideration under § 1.142(b). An applicant who desires examination of an invention so withdrawn from consideration can file a divisional application under 35 U.S.C. 121.
- (1) In an application, other than for reissue or a design
patent, that has been pending for at least three years as of June 8,
1995; taking into account any reference made in the application to any
earlier filed application under 35 U.S.C. 120,
121 and 365(c), no
requirement for restriction or for the filing of divisional
applications shall be made or maintained in the application after June
8, 1995, except where:
- (c) The provisions of this section shall not be applicable to any application filed after June 8, 1995.
“Restriction” under 37 CFR 1.129(b) applies to both restriction requirements under 37 CFR 1.142 and election of species requirements under 37 CFR 1.146.
37 CFR 1.129(b)(1) provides for examination of more than one independent and distinct invention in certain applications pending for 3 years or longer as of June 8, 1995, taking into account any reference to any earlier application under 35 U.S.C. 120, 121, or 365(c). Applicant will not be permitted to have such additional invention(s) examined in an application if:
- (A) the requirement was made in the application or in an earlier application relied on under 35 U.S.C. 120, 121, or 365(c) prior to April 8, 1995;
- (B) no restriction requirement was made with respect to the invention(s) in the application or earlier application prior to April 8, 1995, due to actions by the applicant; or
- (C) the required fee for examination of each additional invention was not paid.
Only if one of these exceptions applies is a normal restriction requirement appropriate and telephone restriction practice may be used.
Examples of what constitute “actions by the applicant” in 37 CFR 1.129(b)(1) are:
- (A) applicant abandoned the application and continued to refile the application such that no Office action could be issued in the application,
- (B) applicant requested suspension of prosecution under 37 CFR 1.103(a) such that no Office action could be issued in the application,
- (C) applicant disclosed a plurality of independent and distinct inventions in the present or parent application, but delayed presenting claims to more than one of the disclosed independent and distinct inventions in the present or parent application such that no restriction requirement could be made prior to April 8, 1995, and
- (D) applicant combined several applications, each of which claimed a different independent and distinct invention, into one large “continuing” application, but delayed filing the continuing application first claiming more than one independent and distinct invention such that no restriction requirement could be made prior to April 8, 1995.
In examples (A) and (B), the fact that the present or parent application claiming independent and distinct inventions was on an examiner’s docket for at least 3 months prior to abandonment or suspension, or in examples (C) and (D), the fact that the amendment claiming independent and distinct inventions was first filed, or the continuing application first claiming the additional independent and distinct inventions was on an examiner’s docket, at least 3 months prior to April 8, 1995, is prima facie evidence that applicant’s actions did not prevent the Office from making a requirement for restriction with respect to those independent and distinct inventions prior to April 8, 1995. Furthermore, an extension of time under 37 CFR 1.136(a) does not constitute such “actions by the applicant” under 37 CFR 1.129(b)(1).
NOTE: If an examiner believes an application falls under the exception that no restriction could be made prior to April 8, 1995, due to applicant’s action, the application must be brought to the attention of the Technology Center (TC) Special Program Examiner for review.
Under 37 CFR 1.129(b)(2), if the application contains claims to more than one independent and distinct invention, and no requirement for restriction or for the filing of divisional applications can be made or maintained, applicant will be notified and given a time period to:
- (A) elect the invention or inventions to be searched and examined, if no election has been made prior to the notice, and pay the fee set forth in 37 CFR 1.17(s) for each independent and distinct invention claimed in the application in excess of one which applicant elects,
- (B) in situations where an election was made in reply to a requirement for restriction that cannot be maintained, confirm the election made prior to the notice and pay the fee set forth in 37 CFR 1.17(s) for each independent and distinct invention claimed in the application in addition to the one invention which applicant previously elected, or
- (C) file a petition under 37 CFR 1.129(b)(2) traversing the requirement without regard to whether the requirement has been made final. No petition fee is required.
37 CFR 1.129(b)(2) also provides that if the petition is filed in a timely manner, the original time period for electing and paying the fee set forth in 37 CFR 1.17(s) will be deferred and any decision on the petition affirming or modifying the requirement will set a new time period to elect the invention or inventions to be searched and examined and to pay the fee set forth in 37 CFR 1.17(s) for each independent and distinct invention claimed in the application in excess of one which applicant elects.
Under 37 CFR 1.129(b)(3), each additional invention for which the required fee set forth in 37 CFR 1.17(s) has not been paid will be withdrawn from consideration under 37 CFR 1.142(b). An applicant who desires examination of an invention so withdrawn from consideration can file a divisional application under 35 U.S.C. 121.
37 CFR 1.129(c) clarifies that the provisions of 37 CFR 1.129(a) and (b) are not applicable to any application filed after June 8, 1995. However, any application filed on June 8, 1995, would be subject to a 20-year patent term.
Form paragraph 8.41 may be used to notify applicant that the application is a transitional application and is entitled to consideration of additional inventions upon payment of the required fee.
¶ 8.41 Transitional Restriction or Election of Species Requirement – pre-GATT Filing
This application is subject to the transitional restriction provisions of Public Law 103-465, which became effective on June 8, 1995, because:
- 1. the application was filed on or before June 8, 1995, and has an effective U.S. filing date of June 8, 1992, or earlier;
- 2. a requirement for restriction was not made in the present or a parent application prior to April 8, 1995; and
- 3. the examiner was not prevented from making a requirement for restriction in the present or a parent application prior to April 8, 1995, due to actions by the applicant.
The transitional restriction provisions permit applicant to have more than one independent and distinct invention examined in the same application by paying a fee for each invention in excess of one.
Final rules concerning the transition restriction provisions were published in the Federal Register at 60 FR 20195 (April 25, 1995) and in the Official Gazette at 1174 OG 15 (May 2, 1995). The final rules at 37 CFR 1.17(s) include the fee amount required to be paid for each additional invention as set forth in the following requirement for restriction. See the current fee schedule for the proper amount of the fee.
Applicant must either: (1) elect the invention or inventions to be searched and examined and pay the fee set forth in 37 CFR 1.17(s) for each independent and distinct invention in excess of one which applicant elects; or (2) file a petition under 37 CFR 1.129(b) traversing the requirement.
Examiner Note:
- 1. This form paragraph should be used in all restriction or election of species requirements made in applications subject to the transition restriction provisions set forth in 37 CFR 1.129(b). The procedure is NOT applicable to any design or reissue application.
803.03(a) Transitional Application — Linking Claim Allowable [R-08.2012]
Whenever divided inventions in a transitional application are rejoined because a linking claim is allowable (MPEP § 809, § 821.04, and § 821.04(a)) and applicant paid the fee set forth in 37 CFR 1.17(s) for the additional invention, applicant should be notified that he or she may request a refund of the fee paid for that additional invention.
803.03(b) Transitional Application — Generic Claim Allowable [R-08.2012]
Whenever claims drawn to an additional species in a transitional application for which applicant paid the fee set forth in 37 CFR 1.17(s) are no longer withdrawn from consideration because they are fully embraced by an allowable generic claim, applicant should be notified that he or she may request a refund of the fee paid for that additional species.
The determination of when claims to a nonelected species would no longer be withdrawn from consideration should be made as indicated in MPEP § 806.04(d), § 821.04, and § 821.04(a).
803.04 Nucleotide Sequences [R-07.2015]
Polynucleotide molecules defined by their nucleic acid sequence (hereinafter “nucleotide sequences”) that encode different proteins are structurally distinct chemical compounds. These sequences are thus deemed to normally constitute independent and distinct inventions within the meaning of 35 U.S.C. 121. Absent evidence to the contrary, each such nucleotide sequence is presumed to represent an independent and distinct invention, subject to a restriction requirement pursuant to 35 U.S.C. 121 and 37 CFR 1.141et seq.
In 1996, the Commissioner of Patents and Trademarks decided sua sponte to partially waive the requirements of 37 CFR 1.141et seq. and permit a reasonable number of such nucleotide sequences to be claimed in a single application. See Examination of Patent Applications Containing Nucleotide Sequences, 1192 OG 68 (November 19, 1996).
In 2007, the Commissioner for Patents rescinded the waiver. See Examination of Patent Applications Containing Nucleotide Sequences, 1316 OG 123 (March 27, 2007). All pending applications are subject to the 2007 OG notice. Note, however, that supplemental restriction requirements will not be advanced in applications that have already received an action on their merits for multiple nucleotide sequences in the absence of extenuating circumstances. For national applications filed under 35 U.S.C. 111(a), polynucleotide inventions will be considered for restriction, rejoinder, and examination practice in accordance with the standards set forth in MPEP Chapter 800. Claims to polynucleotide molecules will be considered for independence, relatedness, distinction and burden in the same manner as claims to any other type of molecule.
See MPEP § 1850 for treatment of claims containing independent and distinct nucleotide sequences in international applications filed under the Patent Cooperation Treaty (PCT) and national stage applications filed under 35 U.S.C. 371.
803.05 Reissue Application Practice [R-07.2022]
37 CFR 1.176 Examination of reissue.
*****
- (b) Restriction between subject matter of the original patent claims and previously unclaimed subject matter may be required (restriction involving only subject matter of the original patent claims will not be required). If restriction is required, the subject matter of the original patent claims will be held to be constructively elected unless a disclaimer of all the patent claims is filed in the reissue application, which disclaimer cannot be withdrawn by applicant.
Restriction practice relating to reissue applications is governed by 37 CFR 1.176(b) which specifies that restriction may only be required between the invention(s) of the original patent claims and previously unclaimed invention(s) set forth in new claims added in the reissue application. The claims of the original patent must not be restricted as being directed to two or more independent and distinct inventions and must be examined together. Where restriction is required by the examiner, the invention(s) set forth by the original patent claims and any newly added claims that are directed to the same invention(s) will be held as constructively elected. Any new claim that is directed to an invention that is independent and distinct from the invention(s) of the original patent claims will be withdrawn from consideration. See MPEP § 1450 for a detailed explanation of this practice. Note that applicant may initiate a division of the claims by filing more than one reissue application in accordance with 37 CFR 1.177. See MPEP § 1451 for a detailed explanation of this practice.
Where a restriction (including an election of species) requirement was made in an application and applicant permitted the elected invention to issue as a patent without filing a divisional application on the non-elected invention(s) or on non-claimed subject matter distinct from the elected invention, the non-elected invention(s) and non-claimed, distinct subject matter cannot be recovered by filing a reissue application. Once an applicant acquiesces to a restriction (including an election of species) requirement, any invention distinct from that elected and prosecuted to allowance—whether originally claimed or not—can only be pursued in a timely-filed divisional application. A reissue applicant’s failure to timely file a divisional application is not considered to be error causing a patent granted on the elected claims to be partially inoperative by reason of claiming less than the applicant had a right to claim. Accordingly, this is not correctable by reissue of the original patent under 35 U.S.C. 251. In re Watkinson, 900 F.2d 230, 14 USPQ2d 1407 (Fed. Cir. 1990); In re Weiler, 790 F.2d 1576, 229 USPQ 673 (Fed. Cir. 1986). See MPEP § 1412.01.

