| CPC A61K 31/48 (2013.01) [A61K 31/137 (2013.01); A61K 31/4045 (2013.01); A61K 31/675 (2013.01); A61P 25/24 (2018.01); G01N 33/6893 (2013.01); G01N 2800/304 (2013.01); G01N 2800/52 (2013.01)] | 31 Claims |
|
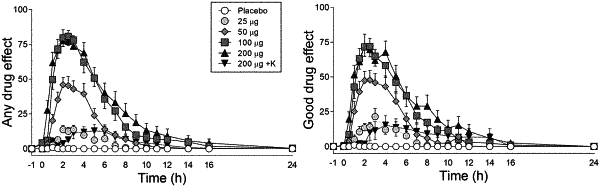
1. A method of dosing and treating patients with a psychedelic, including the steps of:
administering a psychedelic at a dose chosen from the group consisting of a minidose of 21-29 μg, psychedelic dose greater than 30 μg, good effect dose of 30-100 μg, ego-dissolution dose of greater than 100 μg, and cardiovascular safe dose of 50-200 μg, wherein the dose is established through clinical study testing; and
producing maximum positive subjective acute effects that are known to be associated with more positive long-term outcomes.
|